e-DHR stands for electronic Device History Records which maintains the history of products under test. It is widely used in industrial and medical fields where version and data need to be preserved for medical equipment. It requires the products to be manufacturing compliant.
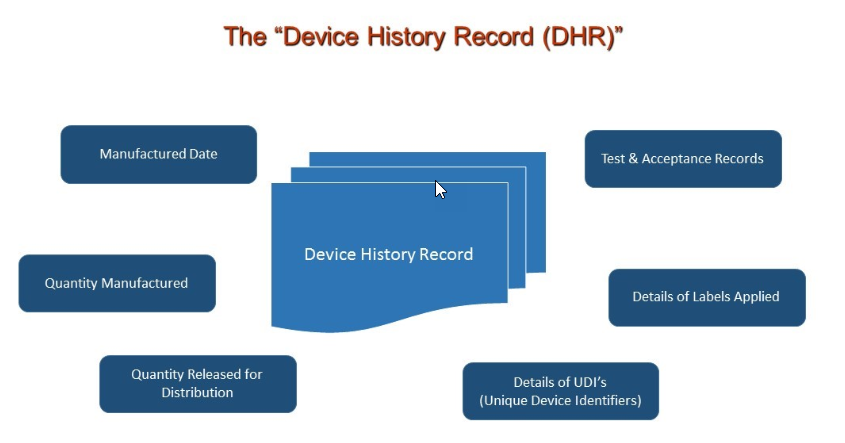
DHR or Device History Record requires following records as a manufacturing compliance. With eDHR the process is automated through Orcanos System for better efficiency and accuracy.