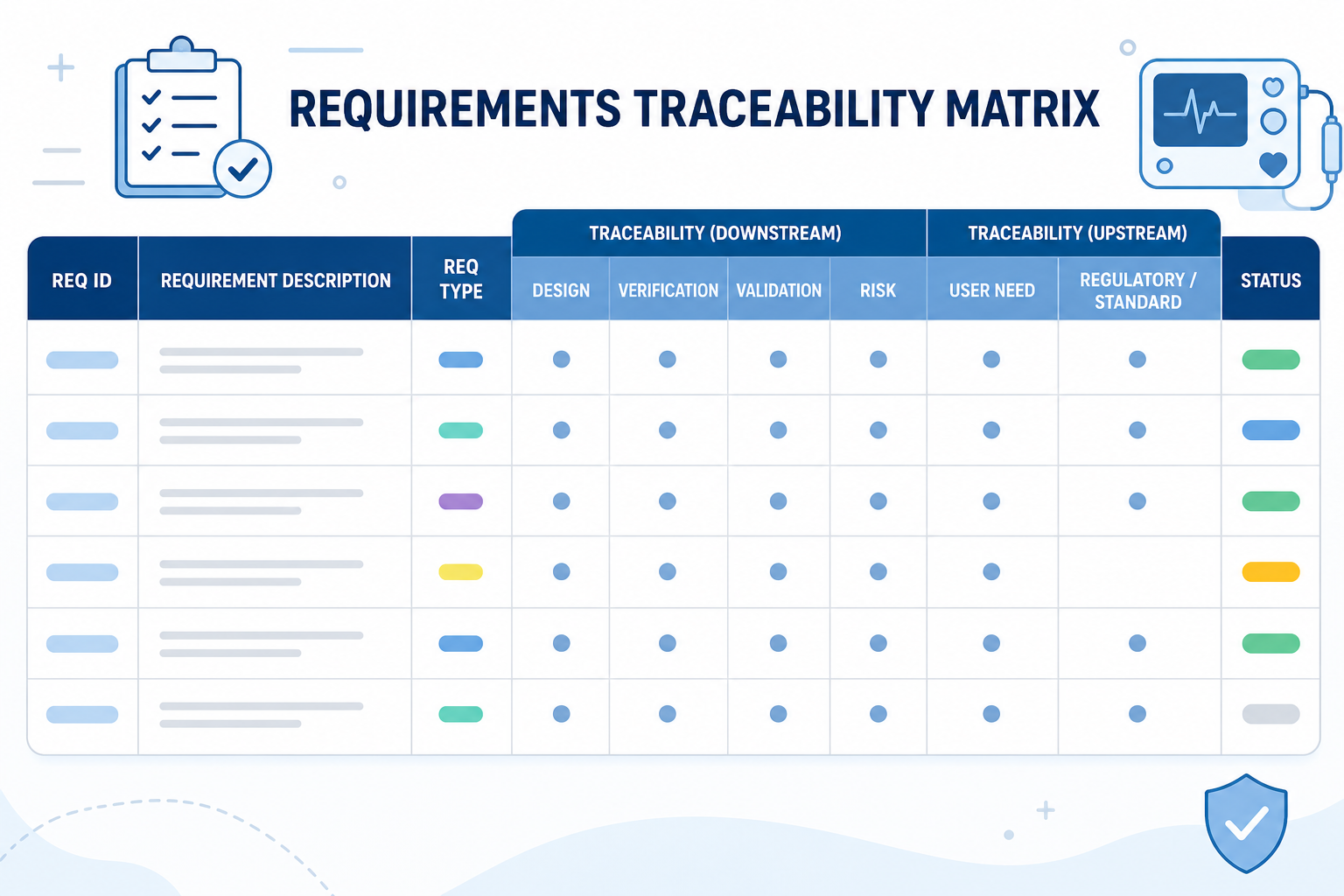
Overview
A traceability matrix is a cornerstone of design control compliance for medical device manufacturers. ISO 13485 and FDA 21 CFR Part 820.30 require establishing clear links between Design Inputs, Design Outputs, Verification & Validation (V&V), and Risk Controls.
Companies risk audit findings, missed requirements, and regulatory noncompliance without a centralized, accurate traceability system.
Migrating from Excel-based traceability matrices to the Orcanos eQMS platform is a major step toward better product lifecycle management. While the transition may appear to reduce the number of traceable items, it actually results in: clearer, more maintainable and meaningful traceability structures. Avoiding unnecessary over-tracing and redundant links between requirements, tests, and risks.
Understanding the V-MODEL in Medical Device Design Control
To align with industry best practices and regulatory expectations, Orcanos supports the widely recognized V-model of design and development.
This model is a common framework in the medical device industry and demonstrates how design inputs are translated into and implemented through design outputs. These outputs are verified and validated against the original inputs, with risk management activities occurring across the lifecycle. This framework is the foundation for traceability in Orcanos, helping ensure that every requirement and risk is addressed through appropriate testing and controls.
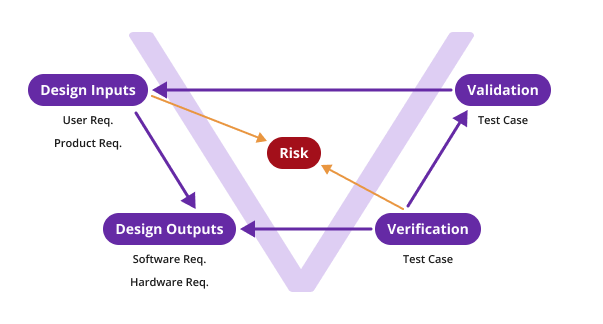
How Orcanos Simplifies Traceability Compared to Excel Sheets
Many organizations that use Excel for traceability create highly detailed Level 4 or Level 5 matrices that link user needs, system requirements, software requirements, design details, and test cases. While this approach aims for completeness, it often introduces unnecessary complexity and effort.
With Orcanos, teams benefit from a streamlined, value-driven approach to traceability that aligns with ISO 13485 and FDA expectations - while keeping the system clear, efficient, and easy to maintain.
✅ Focused traceability that emphasizes meaningful, value-driven relationships
✅ Efficient maintenance with reduced overhead across trace levels
✅ Reliable, accurate links supported by automation
✅ Clear, easy-to-read structure for audits and reviews
✅ Built-in support for effective change impact analysis
✅ Alignment with regulatory requirements without unnecessary trace levels
Orcanos enables teams to concentrate on what truly matters: verifying that design outputs meet inputs, validating that user needs are fulfilled, and ensuring risks are mitigated - all within a single, connected platform.
.png)
Instead of tracking every low-level engineering detail across five columns, Orcanos empowers teams to focus on what matters: verifying that design outputs meet inputs, validating that user needs are fulfilled, and ensuring risks are mitigated - all traceable from a single platform.
This results in a system that’s easier to manage, easier to audit, and more compliant, with less room for error and rework.
Orcanos Traceability Matrix Options
All-in-One Traceability Matrix
The default Orcanos traceability matrix provides a comprehensive view that connects all regulatory-required traceability relationships in a single table:
.png)
This centralized view is ideal for demonstrating full traceability during audits and for performing impact assessments when requirements or risks change.
Optional Breakdown: Separate Verification and Validation Views
For teams or auditors who prefer a more structured approach, Orcanos allows the traceability matrix to be split into dedicated views for verification and validation activities.
This flexibility helps meet different organizational and regulatory reporting preferences, without duplicating data or creating unnecessary complexity.
Validation
Maps design inputs (high-level requirements) to their respective validation test cases, ensuring the final product fulfills its intended use.
.png)
Verification
Connects design outputs (low-level requirements) to their respective verification test cases, ensuring that each implemented feature functions as intended.
.png)
Conclusion
With Orcanos, traceability becomes simpler. It is not about stripping things down, but about helping teams stay focused on what really matters: compliance, product quality, and innovation, without getting buried in manual work.
Traceability does not need to feel messy or overwhelming. When it is structured properly and supported by the right tools, it becomes part of how you work. There is no extra effort and no frustration. Moving from Excel to Orcanos is not only about meeting compliance requirements. It is also about making day-to-day work easier for everyone involved.
A well-built traceability matrix does more than check a box for auditors. It helps teams make better decisions, keeps everyone aligned, and makes sure nothing important slips through the cracks. As regulations continue to evolve, having everything organized in one place is not just helpful, it is becoming essential.
If you would like a personalized demo please schedule here.