Orcanos Document Control: Audit-Ready, Automated, Designed For Medical Device.
Transform manual friction into fully automated system
Orcanos replaces regulatory drag with a 21 CFR Part 11-compliant engine that automates your entire document lifecycle, from initial draft to global distribution


Stop 'Preparing' for Audits. Live in a Constant State of Readiness.
To maintain a constant state of inspection-readiness, the system features an Online Audit module designed to streamline both internal and external evaluations. This functionality transitions the audit process from a reactive, paper-heavy event to a proactive, digital workflow.
Virtual Auditor: A real-time monitoring engine that scans the document database to identify gaps, such as expired SOPs or missing training records, before they become non-conformities.
Instant Traceability: During a live audit, users can instantly pull full revision histories, e-signature logs, and linked CAPAs, eliminating the need to manually search through physical binders.
Integrated Findings Management: Observations and findings are logged directly within the system, automatically triggering corrective actions (CAPA) or document change requests where necessary.
Orcanos was designed with a single mission: to automate the complexity of Medical Device compliance so you can focus on the product, not the paperwork.
If your change control process is in a slow-motion
We replace chaos with compliance. By leveraging Orcanos’ integrated Electronic Change Order (ECO) system, you can enforce structured workflows that route documents for approval instantly. Plus, we fully automate the batch release of related documents, ensuring that every connected SOP, work instruction, or drawing is up-to-date simultaneously, saving weeks of manual effort.
If you still manage spreadsheet-based training matrixes
Orcanos bridges the gap between document revision and training. The moment a new document is approved, the system automatically triggers training tasks for all required stakeholders. There is no manual matrix to maintain. Employees receive instant notifications, can complete training with a click, and the system creates a bulletproof, real-time electronic training record that is always "audit-ready."
When approval workflows are a rigid 'one-size-fits-none' system
We put control back in your hands. You can easily design and implement dynamic, custom workflows that route different types of documents through different approval stages based on their risk or complexity. Whether it’s a simple update or a major revision, Orcanos uses seamless, secure electronic signature modules to move approvals forward instantly and securely from any device.
When the thought of an FDA audit gives you anxiety
We don't just "support" 21 CFR Part 11; we build our entire platform around it. You get absolute certainty that your electronic records, audit trails, and digital signatures meet every regulatory requirement. We provide a validated, compliant environment that provides an immutable history of who did what and when, letting you walk into any audit with confidence.
Secure, Automated PDF Publishing
All approved documents are automatically published as PDFs with a secure watermark and stored indefinitely in a protected vault. When new revisions are released, previous versions are marked as obsolete, ensuring your team always works with the latest information.
Lightning-Fast System Launch
We replace months of implementation with days of action. Leverage Orcanos' "instant-on" platform and effortless migration tools. Your entire existing file repository is centralized and structured, allowing your team to go live with a fully compliant system in less than one week.
Take your regulatory compliance to the next level with Orcanos. Book a demo today and experience the power of our integrated solutions firsthand!

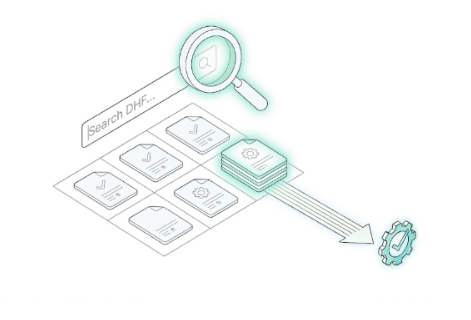
An auditor asks you for the "Design History File for Project X, Revision 2."
Instant Digital Traceability. One search, one click. Every version, every signature, and every related record is linked and retrievable in seconds—no matter where your team is located.
Visual Workflow Dashboards. See exactly where every document is stalled. Automated escalations and "nag" notifications move the process forward without you having to play "document police."

Bulletproof Revision Control—Fully Automated, Fully Compliant
Do you feel like you spend 80% of your time chasing people for signatures rather than improving quality?
Manual revision control is a high-wire act. One missed watermark, an outdated training record, or a "ghost" signature can tank an entire audit.
We can give you your time back:
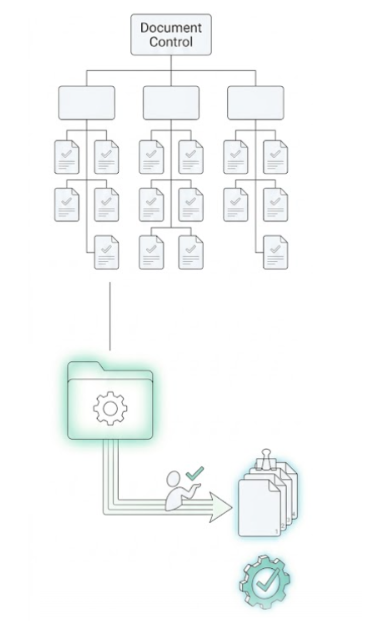
Intelligent Structure & Submission-Ready Export
Most systems are just a "digital dump" of files. You waste hours searching through folders, and when it’s time for a 510(k) or CE Mark submission, the nightmare of manually collecting, naming, and sequencing hundreds of PDFs begins. One missing document can delay your market entry by months.
The Orcanos Solution: We bring logical order to regulatory chaos. Our system allows you to organize your documents in a multi-level, hierarchical tree that mirrors your actual DHF or Technical File structure.
Intuitive Classification: Drag-and-drop documents into a familiar folder-based hierarchy that enforces consistent naming and metadata.
One-Click Submission Export: When you are ready for a regulatory filing, simply select your "Submission Set." Orcanos automatically compiles the latest approved versions into a clean, structured package—complete with a table of contents—ready for the FDA or your Notified Body.
_HighPerformer_EMEA_HighPerformer.svg)
_FastestImplementation_Mid-Market_GoLiveTime.svg)

_HighPerformer_Mid-Market_EMEA_HighPerformer.svg)
.png)
.png)



































