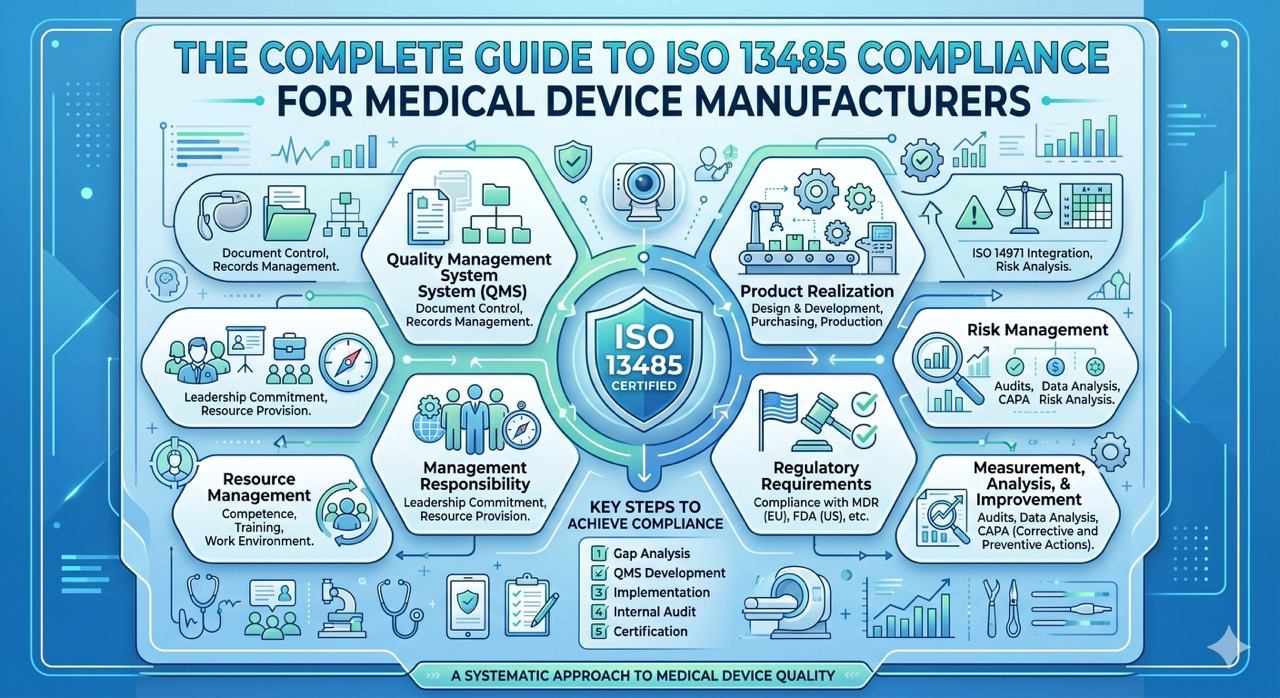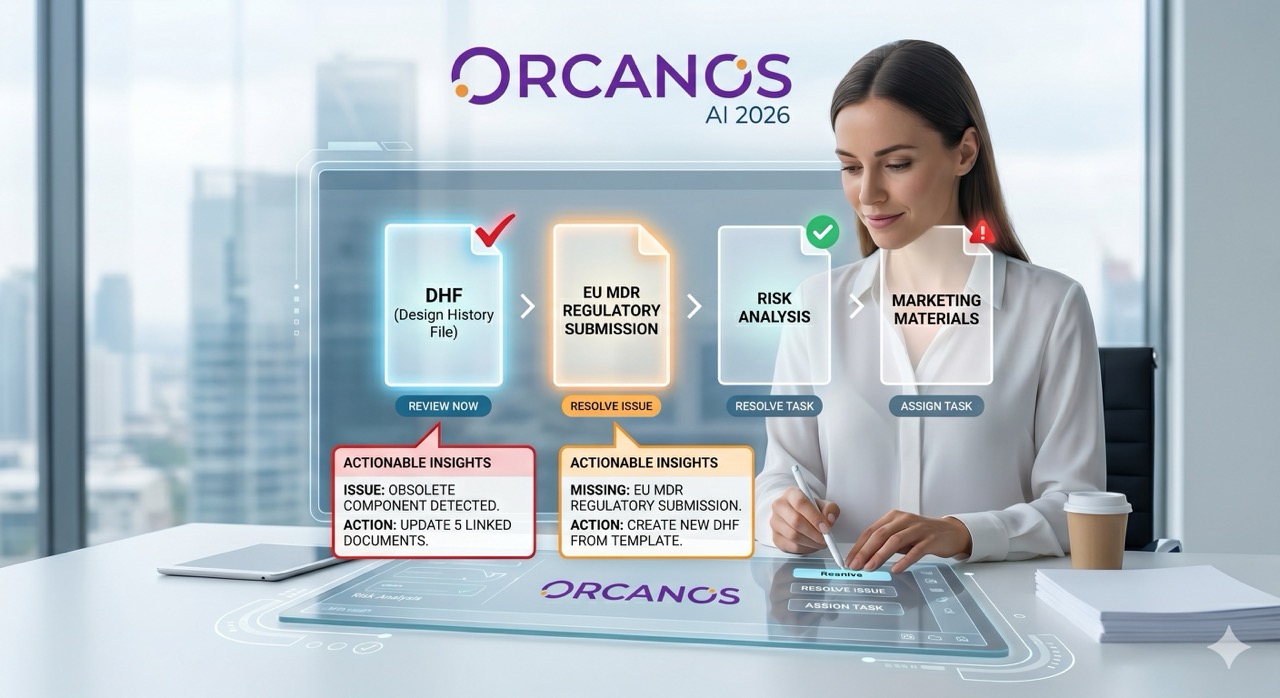
Regulatory audits for Medical Devices and SaMD (Software as a Medical Device) shouldn’t feel like a high-stakes scavenger hunt. Yet, for many teams, the "knock on the door" triggers a wave of panic.
When an auditor asks for proof, you shouldn't have to dig through buried emails or broken spreadsheet links. Orcanos Audit as a Service turns audit anxiety into a non-event by replacing chaos with a structured, digital "Command Center."
The Pain: Why Audits Are Stressful
Most companies struggle during inspections not because they lack quality, but because their evidence is invisible. * Data Silos: Information is trapped in disconnected folders and personal hard drives.
- The "Traceability Gap": Linking a Risk to a Requirement and then to a Test Result takes hours of manual work.
- Human Error: Missing signatures or overdue CAPAs are only discovered when the auditor is sitting in the room.
The Solution: How Orcanos Solves the Audit Puzzle
We don’t just give you software and wish you luck. Audit as a Service is a guided transition from manual "paper-shuffling" to a high-speed digital environment.
1. Cleaning the House (Data Optimization)
We start by scrubbing your current data. Our team helps you remove duplicates, fix broken links, and organize your files logically. You don't move your "mess" into the new system; we optimize it first.
2. Building the "Digital Thread" (Traceability)
In Orcanos, a document isn't just a file, it's a connected object. We link your processes so that one click shows the entire history:
Customer Complaint->Risk File Update -> Design Change -> Verification Test.
3. Automated Guardrails (Workflow Control)
Human error is the #1 cause of audit findings. Orcanos automates your CAPA, NCR, and Change Control workflows. The system ensures that no step is skipped and every approval is timestamped and compliant (21 CFR Part 11).
4. The "One-Click" Evidence Retrieval
When an auditor asks, "Show me all open CAPAs related to this specific risk," you shouldn't need a coffee break to find the answer.
- Audit Dashboards: Pre-configured views for instant reporting.
- One-Click Export: Generate full validation packages or technical files in seconds.
We Stand With You
We don't just set up the software; we support you during the audit. Our experts can assist with system navigation, data retrieval, and configuration adjustments in real-time to ensure the process remains smooth and professional.
Ready for a Stress-Free Audit?
Don't wait for the notification letter to start getting organized. Transition to a system where compliance is a byproduct of your daily work, not a last-minute chore.