.png)
A validation master plan (VMP), which is part of Current Good Manufacturing Practice (cGMP), is designed to help medical device manufacturers by defining, and giving justification for, process validation requirements. If you need to control your processes, or must follow a specific methodology, a validation master plan is important.
Companies specializing in biotechnology, pharmaceutical, and medical device manufacturing must have effective validation of their processes, products, and facilities. This is an integral part of any Quality Management System.
Note that a validation master plan (VMP) differs slightly from a standard operating procedure (SOP). A VMP is used to prioritize objectives and plan the steps required for approved tasks. It is used to capture ALL elements required to support a process, including equipment, benefits, activities, prerequisites, and strategies.
An SOP, on the other hand, outlines the exact procedures to follow to complete approved tasks. Although a validation master plan is not a formal requirement of the FDA Quality System Regulation (21 CFR 820) or ISO 13485, it’s a good idea to have a thorough and effective VMP so that you can be sure you are running an efficient validation program.
What is Validation Master Plan?
A validation master plan, or VMP, records and stipulates methods and equipment that require approval; the reason they are needed; the plan that ensures a particular process is completed; and the person responsible for the validation process.
Creating a VMP will help you avoid trouble in these highly regulated industries. It is not uncommon for FDA auditors to request detailed documentation of procedures, details about equipment, and information about your process validation plans. Despite the lack of formal requirements to have a VMP, having one will make it less likely you’ll be issued a warning letter for failing to produce requested information or documentation with sufficient detail.
Why do we need a Validation Master Plan?
A VMP helps us prioritize activities and predict capital expenditures. It also gives us important insights and helps us create timelines for specific goals. It’s a tool that can be used to track activity, record benefits, monitor prerequisites, and document the approval status of products and equipment. By capturing the full lifecycle of the manufacturing validation process, the VMP works as a project planning tool, ensuring the best overall outcome.
A VMP is also a strategic tool for managing the records for instrument and equipment approval and general procedures. Additionally, the plan can be shown to controllers to demonstrate approval impacts. And finally, it is a great way for medical device manufacturers to show their commitment to, and responsibility for, manufacturing system quality.
What do we need to think about when we create a Validation Master Plan?
Ideally, the VMP should consider risk and the likely impact of the current manufacturing practices on product quality. Using a VMP will help you keep track of which procedures require approval and ensure that validation takes place in a logical order.
The Orcanos Risk Management System helps assess risk and identifies procedures needing approval. This kind of systematic approach is required to survey and determine the effect of each manufacturing process on final product quality. At any point, if you decide that you need any additional prerequisites to the validation, a VMP can be used to capture these extensions or changes.
What does a good Validation Master Plan include?
Although the FDA Code of Federal Regulations does not address any VMP requirements, the Guidance for Industry suggests the following:
- Plans for review
- Plans for any simultaneous validation activities
- Details of the request, timing, scope, and need for validation
- A report explaining the validation approach to all those in the organization
- Details of the timeline, procedures, and facilities
- Departments that have consented to the project
- A description of the validation training program plans
- Specifics about staff approvals for SOPs, the VMP, and surveying and monitoring of tasks
- Copies of all approval plans, reports, and conventions; existing SOPs; strategic planning documents; and any other similar records
What sections should be in a Validation Master Plan?
- Table of Contents: Gives a glimpse into the critical areas of the VMP.
Sample Table of Contents:
- Purpose
- Scope
- Standards, guidelines, and other applicable documents
- Introduction
- Method
- Procedure
- Acceptance criteria
- Responsibilities
- Validation support system
- Process description
- Process validation decision
- Qualification and validation flow diagram
- VMP tracking
- Abutment production flow chart
- List of Appendices
- Authorization and Title Page (dates with approval signatures): Includes a document number, version, title, and management and QA approval. These elements are managed automatically by the Orcanos electronic document management system (eDMS).
- Validation Plan: Identifies why validation is required; what must be validated; and where, when, and how the validation should be done. Also includes a set of procedures broken into sections, highlighting the methods vital to the project.
- Abbreviations and Glossary: Explains any technical and organizational terms used in the plan.
- Purpose and Approach: States the purpose of the chosen validation approach. Remember that this document should be both detailed and concise. It must be clear and easy to read and you must keep the end user in mind when you write it. The VMP outlines the framework, procedures, equipment, and planned execution of the approach. Even when there’s a change in control and capability, the framework and approved method assures all products follow a well-documented risk evaluation assessment so that integrity remains in the quality management system and the product.
- Scope: Covers all utilities (electricity, method etc.), systems, processes, procedures, and equipment specific to the manufacturing process. Based on the documented risk evaluation, the scope must capture the framework, procedures, utilities, and strategies that will be validated. The scope must also define who is responsible for which elements of the plan. Like all process documents, it is crucial that the scope is clear to all readers.
- Outsourced Services: Outlines which services relating to the management and selection of qualifications, calibrations, and activities will be performed by a third party.
- Roles and Responsibilities: Details who does what, in terms of the various elements of the plan: preparing validation protocols; managing change control documents and tasks reports; validating SOPs; maintaining and storing all validation related documents. The VMP will be referenced frequently throughout manufacturing, approvals, reporting, and validation so the roles and responsibilities need to be clearly stated and easy to read.
- Deviation management: Outlines the method for documenting deviations. Note that when a deviation occurs during approval, a determination of a suitable corrective action is made. The Orcanos electronics CAPA management system is the ideal tool to support this process.
- Risk management: Includes the risk management principles used in the validation process. Risk assessment must include considerations relevant to design, manufacturing, distribution,, and the entire lifecycle of a planned project. The Orcanos risk management system ensures risk is managed according to the requirements of ISO 14971:2019.
- Change Control: Reflects the change management (ECO) system, which must be in place to effectively manage all changes to the validated processes.This can be easily managed using Orcanos ECO management software.
- Training: Defines personnel involved in a project, the required qualifications, and the training needs. Orcanos training management system is an effective tool for ensuring timely and complete training of all staff involved in a project.
- Validations: Clarifies all elements requiring validation including: the analytical method, cleaning, processes, equipment, premises, computer validation, qualification, utilities, and any revalidation requirements. The Orcanos validation management system ensures that you’ve got everything covered and well monitored and managed using Orcanos V&V module.
The VMP must always include details about critical areas such as manufacturing, central plant, and material storage. It’s also critical that connections between elements of the plan are clear and illustrations and visual aids are used where required for the sake of clarity. A VMP may also need to differentiate between regions, for instance non-GMP versus GMP regions. And finally, a VMP needs to consider and include general acknowledgment criteria of the validation results as well as any advances in hardware, procedures, frameworks, or any other relevant considerations.
- Validation Matrix: This provides a snapshot of all the necessary validations, and their interactions, throughout the project. By using a matrix which connects all the elements, timelines for crucial approvals will always be met.
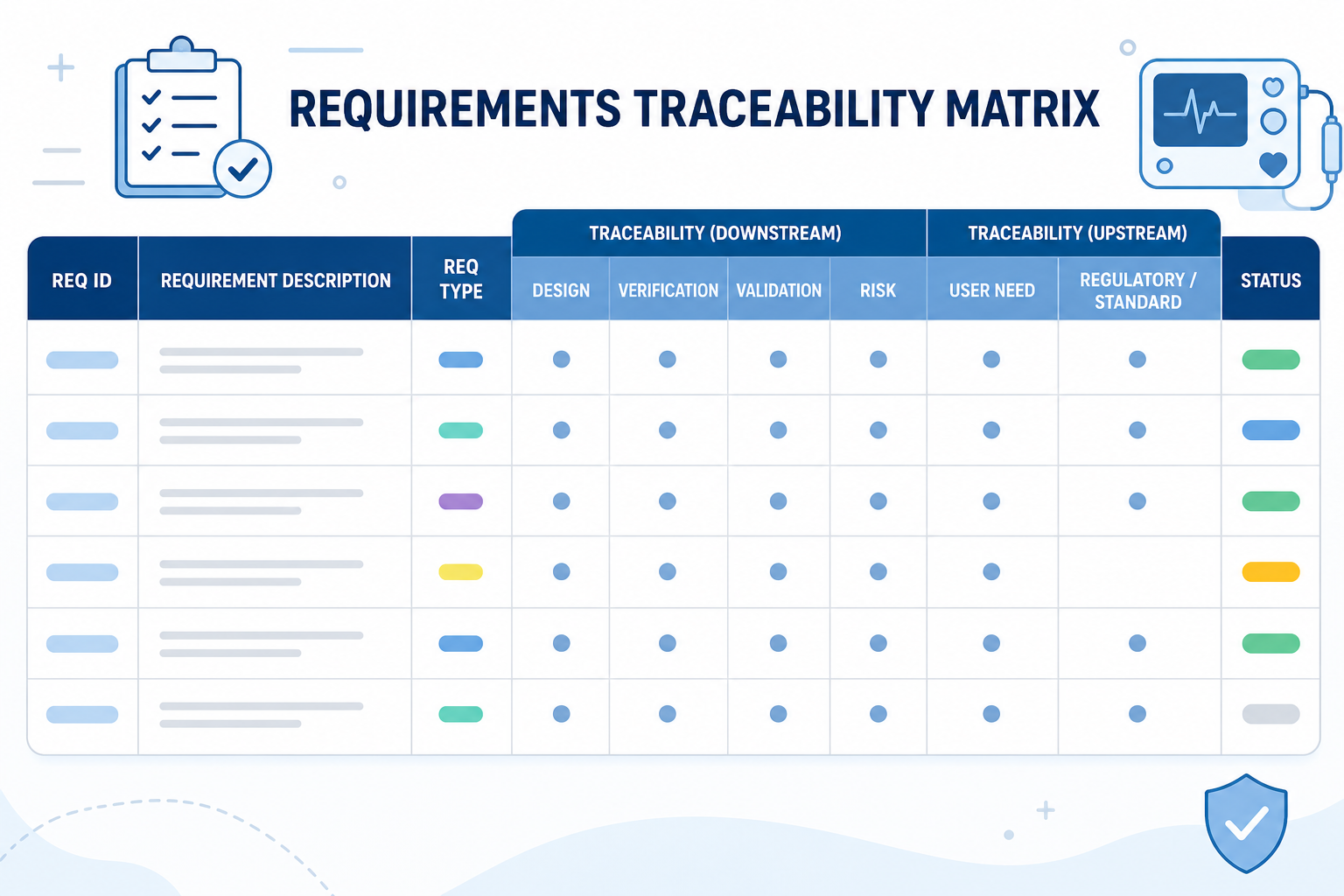
- References: This captures, and makes it easy to find, records of all required activities, directions, executions, approvals, actions, and capabilities in the VMP and is easily done using Orcanos traceability tools.
Quality Assurance is a top priority for everyone in the medical industry. A validation master plan (VMP) is a critical piece of the puzzle when it comes to ensuring the level of quality that we strive for. As part of our Orcanos system and services, we help you manage Operational Qualification (OQ), Installation Qualification (IQ), and Performance Qualification (PQ) protocols. Reports from our system can be generated in any format, using any data settings. The Orcanos eGMP provides flexible, full-scale capabilities to allow any organization to create and manage an effective validation master plan. The Orcanos electronic forms (eForms) combined with built-in process automation infrastructure can increase your organization’s efficiency by over 25%. More importantly, it can reduce risk during the production process and ensure you have processes and systems with integrity and a product you can be proud of.
To learn how we can help you make a difference in this field, and be recognized for the quality of your organization and product, please reach out, we’d love to help.