.png)
Introduction
Looking to expand your knowledge of medical devices? Then, this white paper is for you to read. During this paper, you will learn the primary concepts of design control as well as the necessary things needed in order to be successful in product development of medical devices such as –
- Intended use
- User needs
- Design input
- Design verification
- Design validation and
- Design transfer
Goals You Can Achieve
Listed below are the goals to be achieved with this short course
- To get a basic knowledge of design control for medical devices and why it is important.
- To figure out more specifically how to implement design control in the Orcanos ALM system for design control. Orcanos offers on www.orcanos.com vas knowledge for the Medical Device and Automotive industry at the same time. This could be useful for your job or career
Orcanos.com also offers a full eQMS cloud base platform for complimentary quality procedures and processes used by regulated industries such as Medical Device, Automotive which is quite similar to this one but much comprehensive with more in-depth requirements.
By the end of the paper, you would be equipped with knowledge that might prove to be very useful with Auditors.
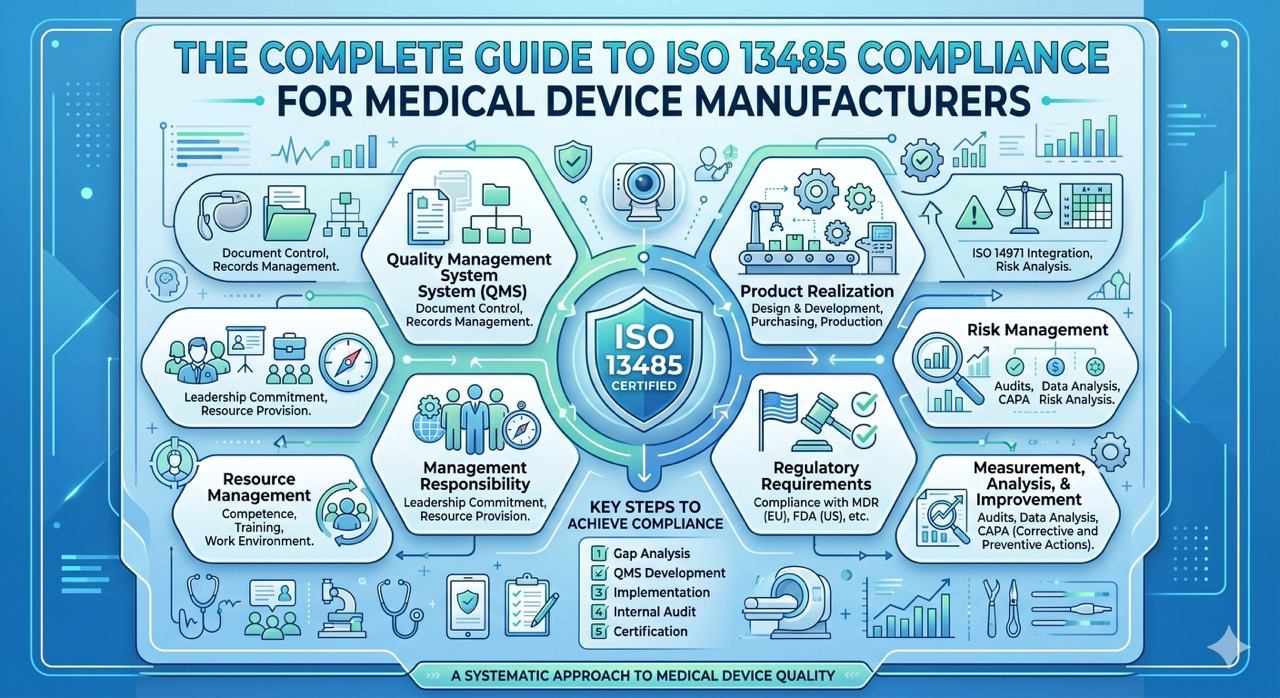
What is Design Control?
Design control is a quality system regulation that covers requirements on how to carry out product development of a medical device or basically the name of chapter 21 of CFR 820.
Understanding design control requirements is necessary especially when selling to the United States because design control requirements are mandatory for medical devices in the US. It goes as much to say that no design control also means no market access.
The same requirements also apply if you’re selling to the European Union but instead they can be found in chapter 7.3 of the EN 1S0 13485 standard
Why are design controls important?
Here are 2 good reasons why you should care about design control –
1. Regulatory Requirements
Conforming to design control requirements is a regulatory requirement and working according to these requirements is an effective way of maintaining the production of safe products that will satisfy your customer’s needs.
2. Best practices
The design control requirements also represent best practices in developing new products and taking a look at companies from other industries where design control requirements are not necessary, you would still find them abiding by these principles anyway because they recognize the values behind it.
Learning about design control is not only about complying with certain requirements in order to gain access to the US and European Markets. Its major purpose is to help develop successfully safe products that also meet your customer’s needs.
It’s important to understand the industry language of whatever industry you work in and the same also applies to Medical device product development. If you happen to work in other product development industries before joining medical devices you’ll find that many concepts are the same but there are 2 things that are essentially different. First is that there are different names and terms for things and secondly the obsession of Risk management and Safety.

Because of that unique link between the design control and RISK not many systems allow managing both in the same system. Orcanos provides a unique ALL IN ONE platform over the cloud that interconnects these elements as creating a collaborative environment to streamline data from design control to risk and vis versa.
In this short paper, we would be bringing some of the popular keywords that are used very often to your attention. Getting to know the medical device terminology will prove very useful especially with understanding your colleagues and your company’s quality procedures.
If you want to learn more about the things we will be sharing information on our blog discussing on to orcanos.com
Principles of Medical Device Product Development
Intended use/Intended purpose
Intended use is a short but high-level description of the user profile, what indications the device is intended for, patient groups and other important conditions of use as intended by the product manufacturers.
This is a term that you should have defined quite early at the beginning of a project. For better understanding, here is an example of the intended use for a thermometer – which is so commonly used nowadays during the COVID-19.
ThermoScan Thermometer
The Thermoscan thermometer is designed for intermittent measurement and monitoring of human body temperature for both adults and children. It is intended for household use only.
It is important to get the intended use of a product correctly because in nations like the US, whether the product will be cleared under a 510k or require pre-market approval depends on the Intended use.

A simple way of coming up with your intended use is to answer important questions like the 5W technic.
- Who
- Whom
- What
- Where
- When
For instance, who would be using the device and on whom for, what where and when is the device used. The trick is to answer these questions using simple sentences and you’ve successfully defined your intended use.
Sometimes, not all intended uses will be fully defined and properly answered but if you can manage to answer them all then you should be able to properly describe what your intended use should be like.

User needs
User needs are another important concept you should be defining at the beginning of the project. As the name implies your user needs should simply define the needs of the users and they must be in line with the intended use. An example of user needs is that the system should be portable
Design Input
These are an abstract technical requirement that realizes the user needs when implemented in the design. The user needs is what is being translated into design input.
For instance, if the user needs to require that a system should be portable and in this case, portable can mean a lot of different sizes for the user. We can decide that the weight of the system should not be more than 10kg and another person might choose to go with another weight that might also work out for this particular case.
At the end of the day, what we try to do is to come up with abstract requirements that would work best for the users according to the user needs. Getting the translation for user needs into the right technical requirements is very important for consumer acceptance as well as the overall success of your product.
There is no part of the regulatory requirements that indicates who should come up with the user needs and design inputs but most times the people who shoulder the responsibility of defining user needs are usually found in Management or Product Management and Marketing. Orcanos system supports all types of design inputs and if needed more types the enterprise edition allow you to add more requirements as needed.
If the design input is technical in nature they are usually established by the engineering department and would feature engineering language.
In some cases, the design inputs may act as the mitigation for the RISK introduce by the user requirements or in other cases they can act as the source for RISK as well. This all depends on the size of the organization or the complexity of the device.
Design Verification
It’s a general principle in Product Development that after designing a product, you have to check that the design or product you came up with actually correlates with the requirements you defined at the beginning of the project. For the Medical Device Industry, this process is an important regulatory requirement termed Design Verification
Design verification confirms that design output meets design input requirements. In this scenario, the design input was that the device weight should not be more than 10kg and to verify that this requirement adhered to, the device would be placed on a calibrated scale and weighed accordingly.
There a lot of methods and processes that can be used for verification and demonstrating that your design output has met the requirements of your design input and in most cases, the methods or process you use is up to you to decide on.
After confirming that the product meets the necessary technical requirements, you also have to check that the device is in due accordance with the intended use and user needs.
Design Validation
In real-life settings, sometimes a product can be 100% technically accurate but still very much useless. To avoid that the device has to undergo a design validation process.
Design Validation is another important regulatory requirement and it must be done by testing your device or design in a real or simulated environment. According to the requirements, you also have to use production units or equivalent devices during the process.
Still using the example mentioned above – with a product that is required to be portable you could let a few users carry the device for short while. Using their responses you can thereby conclude if the product is portable or not.
Although, it’s not every time that Design Validation is that simple because sometimes you cannot confirm that the intended use and user needs have been met without carrying out a clinical investigation or use on real human subjects
Competent Authorities
There are various regulatory bodies across the world that ensures that medical devices companies are doing things the way they should be done. For instance, each country in the EU has a supervising government body for medical devices. In the United Kingdom that would be the Medicines and Healthcare Products Regulatory Agency (MHRA). In Sweden and Denmark we have the Medical Products Agency and the Danish Medicines Agency respectively. We also have the Food and Drug Administration (FDA) for the USA. In general, we call these organizations Competent Authorities.
Notified Bodies
In most cases when you sell to the European Union, you would never have to contact or meet with the competent authorities except something goes terribly wrong with the medical devices. Because the auditing of medical device manufacturers, for most manufacturers selling to the European Union is done by companies that have earned the status of being notified bodies.
The notified body assigned to you will come to audit you periodically throughout the year depending on your product and audit scheme and since you are their customer you would be charged a fee for these audits.
You will be communicating with the notified bodies and the FDA to receive your market clearance and approval. For countries In the EU, that would be receiving the CE mark but for countries in the US, you would receive an FDA clearance or approval depending on which classification your product has and which market access route you have taken.
Next, we would be looking at how this design control terminologies are utilized in a product development process.
Products Development Proccess
The following steps are involved in the products development process –
Initiation
Starting a project formally is referred to as Initiation. Initiation should or could be a fairly short phase involving just a few days to make the decision, to begin with. The decision may also be well documented in a project charter.
From a regulatory point of view, Initiation does not necessarily require any formal requirements but most times some Auditors or Inspectors might ask you when the project actually started and the initiation and signing of a project charter would be a great way to show just that.
Planning
Some people might think that it’s hard to get the planning started or maybe there is still too much uncertainty to create a relevant plan. This is where it might be very wise to consider running a pre-study before you start planning.
There are many different names for this phase. It could also be called a feasibility study or concept study but whatever the name is its purpose is to remove doubts by decreasing uncertainty to a level where it is appropriate to start the project under design control.
Good piece advice is to not wait too long before getting the planning started this is a common mistake at this phase of the project.
There are 4 questions you’ll need to ask yourself during the planning phase which are what, when, who and how much.
The first question to ask yourself when you start planning is what? What is the purpose of this project or what objectives are you trying to obtain? The answer to this should contain the intended use, user needs and the design input that we’ve mentioned previously during the course.
In project management terminology this can be likened to defining the scope of the project as well as the deliverables and the product requirements.
People in your organization would usually expect the project manager to come up with who will be doing the work and when they will be doing the work. In many cases, the latter would be documented in a Gantt chart. This is another tool Orcanos provide you to allow you to control and govern the project
Traditionally, the planning we have talked about so far would be carried out as part of a typical project management but from a design control point of view it is seen as a major requirement and is referred to as design planning.
Lastly you should define how much the project would cost. The FDA and other notified bodies do not necessarily have any involvement with how much your projects should cost which is why having a budget is not a regulatory requirement as long as the devices meet up with the proper requirement in terms of safety and efficacy
Don’t worry if you’re not familiar with the terms we’ve discussed from your own processes and procedures. Most times different companies use different names. As a general approach we try as much as possible to stick to terms from standard and norms or what is most commonly used by the industry.
Execution and Design
The next step after planning is execution and design and it involves 3 phases
- Design
- Design verification
- Design Transfer
Design
If you ask anyone the easiest part of the project to relate with is design. It is quite simply the phase that is focused on creating the design and it’s usually the first thing to do in the execution phase of a product development project
For example design would be to design parts and select components for a mechanical device and for electronics it would be creating schematics, making circuit board layouts and selecting components. Design for software would mean to create the code. For a complex system design would mean to do everything above mentioned as well as integrating them as a system.
Design verification
A device should be designed based on the design input that was defined during the planning phase and when the product has been designed you would have to show that what you did meet with the requirements of the design inputs and this process is known as design verification.
During Design Verification you should be able to provide objective evidence that your design outputs which the design you have created is in line with the design inputs or otherwise known as the product requirements. A good way to prove this is to test the device.
You may do design verification on early prototypes based on a regulatory point of view but you have to be wary because it’s possible that the products you create during your first production might be different from your early prototypes.
In most cases, the early prototypes from the engineering department perform better when compared to the products that eventually come out of the first production. This usually happens because you had your best engineers build and tweak the first prototypes until they did what they were created to do and when these engineers are not around during production you might end up with different results at the end of the day. Orcanos will provide you with an eDHR system that will make sure that your Design inputs are tested during the QC of your production.
This brings us to another important process which is creating the capability to manufacture the device in production.
Design Transfer
There should be a part of your plan dedicated to setting up production for your medical device. It is not just good advice but it’s also a regulatory requirement and that important element of product realization should not be left to chance. This process is usually referred to as design transfer
Design transfer comprises of areas such as
- Supplier evaluation
- Designing the manufacturing process
- Buying tools
- Performing process validation and
- Acceptance by manufacturing
Some companies might refer to design transfer and reserve as a ramp-up for production after regulatory approvals and there is nothing wrong with that since there is no rule that requires you to call certain phases by certain names. However, it does help to use commonly used names because it facilitate easy communication with regulatory organizations such as your notified body and the FDA.
Design Validation
After proving that your device is technically alright with the design verification and setting up production the next step is to of course validate the design.
The design validation should confirm that user needs have been met and that your device works like it is intended to. Please do not confuse design validation with process validation or usability testing because they are not the same thing.
Close-out
It’s possible for you to close down the project if you are not the project manager or not involved in clinical investigations or also regulatory submissions. This would be perceived as an administrative closure of the project involving nothing required from a regulatory point of view.
Clinical investigation
It’s a very good idea to learn about what worked and what didn’t work in your project. You can spend a few days ruminating on what you’ve learned and when you are done you could either CE mark the device or proceed to carry out a clinical investigation and then CE mark the device but for the US market that would be seeking for an FDA clearance or approval via a premarket approval process or pre-market notification.
Some companies use a phase call Post Marketing Survalinace (PMS) to understand better how the product performs. This phase means the collection of complaints from the customers and analysis of theses complaints and performs preventive measures. Part of the Orcanos eQMS solution includes a complete complaint management system to comply with the PMS requirements guidelines, given by the ISO 13485 Sec. 8.2.
Production
Finally, you’re at the end of the project and you’ve handed over the necessary responsibilities to the production department. You should still work with design control so if there is a need for any change during this phase you can implement change control on the product. The Engineering Change Order (Notification) called ECO/ECN process involves the ability to identify each change and to asses each change against the potential RISK it may introduce to the patient/user/operator/doctor. Orcanos includes a complete ECO system that allows the manufacture to execute the ECO procedure with 100% accuracy.
You might also be need to update risk management file and clinical evaluation report as well as other relevant documents as you learn from your products on the market.
We have introduced you to a whole lot of design control requirements and principles together with the project process and we hope that by now you have a better understanding on the basic concepts of design control
If you need templates for more inspiration or knowledge on design control, check out our free evaluation system which includes templates on product development at orcanos.com or you can also consider taking the time to meet with us using our online scheduling link.